Views: 0 Author: Site Editor Publish Time: 2025-03-07 Origin: Site










Nylon, a type of polyamide (PA), is a synthetic polymer widely used across various industries, including textiles, automotive, aerospace, and electronics. It is highly valued for its strength, durability, wear resistance, and versatility. In this article, we will explore the structure of nylon, the raw materials used in its production, and its historical development, offering a detailed understanding of its significance in modern industries.
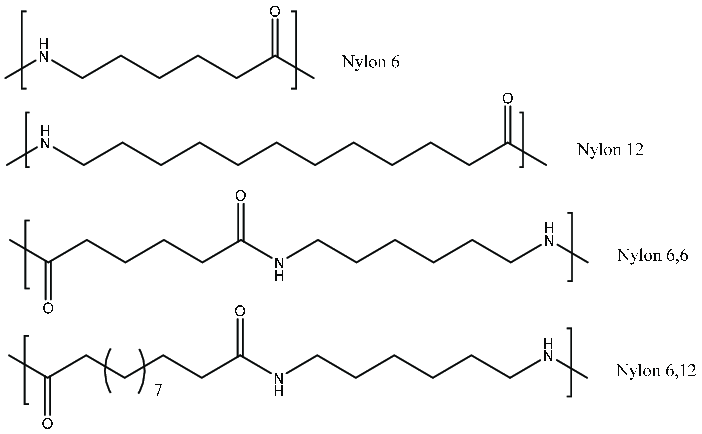
Nylon is a polymer made up of repeating amide bonds (-CONH-). These bonds are formed through a condensation reaction between carboxyl (-COOH) and amine (-NH₂) groups, linking polymer chains together. Depending on the molecular structure and the length of the carbon chains, different types of nylon are created, each offering distinct properties suitable for various applications.
Common types of nylon include:
PA6 (Nylon 6): Made from caprolactam, this nylon type is highly flexible, with excellent wear resistance and tensile strength. It is widely used in fibers and industrial components.
PA66 (Nylon 66): Formed from hexamethylenediamine and adipic acid, PA66 is stronger and more rigid, providing superior heat resistance.
Other nylons: Variants like PA11, PA12, and PA46 offer specialized properties such as high temperature resistance, chemical resistance, and lightweight performance for niche applications.
The structure of nylon contributes to its high mechanical strength, abrasion resistance, and thermal stability, making it an ideal material for both fibers (used in textiles) and engineering plastics (used in automotive and industrial applications).
Nylon production begins with monomers, which are chemicals that can be polymerized to form long chains of molecules. The key raw materials used to create nylon include:
Caprolactam: Used to produce Nylon 6. This cyclic lactam is polymerized to form a long, flexible polymer chain.
Hexamethylenediamine and Adipic Acid: These two monomers are combined to form Nylon 66, which has a more rigid molecular structure and offers greater strength and heat resistance.
Ricinoleic acid-based lauryl lactam: Used to produce Nylon 11, a more eco-friendly, bio-based version of nylon.
These raw materials undergo polymerization or condensation reactions to form long chains of polyamide molecules, which then undergo processes like spinning (for fibers) or injection molding (for engineering plastics) to create finished products. The properties of the final nylon product depend largely on the specific monomers and polymerization methods used.
While traditional nylon production relies on petrochemical feedstocks, the development of bio-based nylons (such as PA11) is helping make nylon more sustainable by reducing dependence on fossil fuels.
The history of nylon is a story of scientific breakthrough, commercial success, and industrial transformation. First synthesized in the 1930s, nylon revolutionized the textile industry and later impacted engineering materials. Below is a brief timeline of nylon’s historical milestones:
1930s: Invention and Breakthrough
Nylon was first synthesized by Wallace Carothers and his team at DuPont in 1935, becoming the first synthetic fiber made entirely from petrochemical sources. In 1939, DuPont introduced nylon stockings, which quickly gained popularity due to their strength, elasticity, and durability. Nylon’s early success in textiles marked a turning point in the development of synthetic fibers.
petrochemical sources. In 1939, DuPont introduced nylon stockings, which quickly gained popularity due to their strength, elasticity, and durability. Nylon’s early success in textiles marked a turning point in the development of synthetic fibers.
1940s-1960s: Widespread Use and Expansion
During World War II, nylon found critical applications in military gear, including parachutes, ropes, and tents. Its high strength and wear resistance made it ideal for these demanding uses. After the war, nylon’s commercial use expanded into consumer goods, particularly clothing and textiles. In the 1950s and 1960s, Nylon 6 and Nylon 66 were developed as engineering plastics, leading to their use in mechanical parts, automotive components, and electrical equipment.
1970s-Present: Diversification and Technological Advancements
As chemical technology advanced, new forms of nylon emerged, including Nylon 12, Nylon 46, and semi-aromatic nylons. These newer nylons offered improved chemical resistance, high-temperature stability, and lightweight properties. Additionally, modified nylons—such as glass fiber-reinforced, flame-retardant, and self-lubricating nylons—became widely used in industries like automotive, electronics, and high-tech manufacturing.
In recent years, the development of bio-based nylons (such as PA11 and PA12) has further contributed to sustainability, reducing nylon’s environmental footprint by using renewable plant sources instead of petroleum-based feedstocks.
Nylon, as a polyamide polymer, has played a central role in advancing the fields of textiles, engineering plastics, and automotive parts. From its early use in fashion and military gear to its current applications in industrial and high-tech sectors, nylon’s structural composition, raw material sources, and historical development demonstrate its ongoing significance in modern manufacturing.
Nylon’s diverse properties, including high strength, abrasion resistance, flexibility, and chemical stability, make it one of the most versatile synthetic materials in use today. With continuing advancements in sustainability and material science, nylon will remain an essential component of various industries for the foreseeable future.
content is empty!
content is empty!


